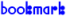N-Methylethanolamine
| Molecular formula: | C3H9NO |
| Molar mass: | 75.11 g·mol-1 |
| CAS registry number: | 109-83-1 |
|
H. Haider, R. Yusoff and M. K. Aroua Equilibrium solubility of carbon dioxide in 2(methylamino)ethanol Fluid Phase Equilib. • Year: 2011 • Volume: 303 • Pages: 162-167. Keywords: Absorption, Alkanolamine, 2(Methylamino)ethanol, CO2 solubility, Equilibrium constants DOI: 10.1016/j.fluid.2011.01.016 ThermoML: http://trc.nist.gov/ThermoML/fpe/j.fluid.2011.01.016.xml |
|
Alvarez, E.[Estrella], Gomez-Diaz, D.[Diego], Navaza, J. M.[Jose M.], La Rubia, M. D. Surface Tension of Aqueous Binary Mixtures of 2-(Methylamino)ethanol and 2-(Ethylamino)ethanol and Aqueous Ternary Mixtures of These Amines with Triethanolamine or N-Methyldiethanolamine from (293.15 to 323.15) K J. Chem. Eng. Data • Year: 2008 • Volume: 53 • Pages: 318-321. DOI: 10.1021/je700536m ThermoML: http://trc.nist.gov/journals/jced/2008v53/i01/je700536m.xml |
|
S. Kapteina, K. Slovik, S. P. Verevkin and A. Heintz Vapor Pressures and Vaporization Enthalpies of a Series of Ethanolamines J. Chem. Eng. Data • Year: 2005 • Volume: 50 • Pages: 398-402. Keywords: vapor pressure, enthalpy of vaporization, hydrogen bond DOI: 10.1021/je049761y ThermoML: http://trc.nist.gov/journals/jced/2005v50/i02/je049761y.xml |
|
Venkat, A., Kumar, G., Kundu, M. Density and Surface Tension of Aqueous Solutions of (2-(Methylamino)-ethanol +2-Amino-2-methyl-1-propanol) and (2-(Methylamino)-ethanol + N-Methyl-diethanolamine) from (298.15 to 323.15) K J. Chem. Eng. Data • Year: 2010 • Volume: 55 • Pages: 4580-4585. DOI: 10.1021/je1002626 ThermoML: http://trc.nist.gov/ThermoML/10.1021/je1002626.xml |
|
K. Klepacova, P. J. G. Huttenhuis, P. W. J. Derks and G. F. Versteeg Vapor Pressures of Several Commercially Used Alkanolamines J. Chem. Eng. Data • Year: 2011 • Volume: 56 • Pages: 2242-2248. Keywords: vapor-liquid equilibria (VLE), vapor pressure, alkanolamine, ebulliometer DOI: 10.1021/je101259r ThermoML: http://trc.nist.gov/ThermoML/10.1021/je101259r.xml |
|
Li, J., Mundhwa, M.[Mayur], Tontiwachsuthikul, P.[Paitoon], Henni, A.[Amr] Volumetric Properties, Viscosities, and Refractive Indices for Aqueous 2-(Methylamino)ethanol Solutions from (298.15 to 343.15) K J. Chem. Eng. Data • Year: 2007 • Volume: 52 • Pages: 560-565. DOI: 10.1021/je060457+ ThermoML: http://trc.nist.gov/journals/jced/2007v52/i02/je060457+.xml |
|
M. Mundhwa and A. Henni Molar Heat Capacity of Various Aqueous Alkanolamine Solutions from 303.15 K to 353.15 K J. Chem. Eng. Data • Year: 2007 • Volume: 52 • Pages: 491-498. DOI: 10.1021/je0604232 ThermoML: http://trc.nist.gov/journals/jced/2007v52/i02/je0604232.xml |
|
M. Mundhwa and A. Henni Molar excess enthalpy oHE mP for various {alkanolamine (1) + water (2)} systems at T = (298.15, 313.15, and 323.15) K J. Chem. Thermodyn. • Year: 2007 • Volume: 39 • Pages: 1439-1451. Keywords: Molar excess enthalpy, Aqueous alkanolamine, NRTL, UNIQUAC, UNIFAC DOI: 10.1016/j.jct.2007.03.010 ThermoML: http://trc.nist.gov/ThermoML/jct/j.jct.2007.03.010.xml |